
Seeking patients ages 12 and older with PSC to learn if the investigational medication, volixibat, might be an effective and safe treatment option for itching due to PSC.



Additional study requirements and exclusions apply. A study representative will discuss these with you during the screening period.

PSC is a rare liver disease that causes damage to the bile ducts inside and outside the liver. It is frequently associated with inflammatory bowel disease (IBD).
A common complication from PSC is itching which can lead to fatigue, social isolation, and an overall lower quality of life. Currently, there are no approved or effective medications to treat PSC or PSC-associated itch.
Here is a helpful breakdown of what will take place during the study, which will last at least 32 weeks (~8 months) and the Optional Long-Term Extension (LTE).
*For USA only: you may be eligible to complete all visits from home.
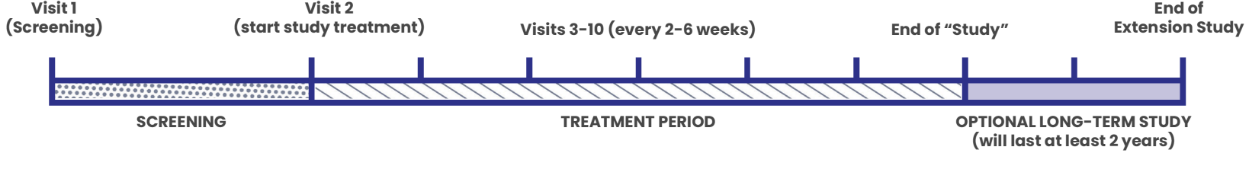

Study staff will assess your itch and your health and determine if you are eligible for the VISTAS Study. Upon entering the screening period, you will need to record your itch score once each day. Recording your itch score will take less than 2 minutes.

You will be randomly placed on volixibat or placebo, which you will take twice a day by mouth. The study team will perform study related tests and procedures and ask questions. Some visits can be performed virtually or remotely. You will be asked to continue recording your itch score once each day.

Every patient, regardless of whether they were on volixibat or placebo during the treatment period, will be guaranteed to receive volixibat and be monitored by the study doctor for at least 2 years. Participants will be asked to continue recording their itch score once every day.
A clinical trial tests the safety and effectiveness of an investigational medication in human volunteers. Every investigational medication goes through the clinical trial process. Therefore, participants play a vital role in advancing medicine for present and future generations
An investigational medication has not been approved by the U.S. Food and Drug Administration (FDA) or other regulating body but is allowed to be administered to people for research purposes. Volixibat is an investigational medication currently being studied for treatment of PSC.
A placebo has no active ingredient and is used to compare the effects of the investigational medication. The placebo will look like the investigational medication and will be administered the same way.
Agreeing to join a clinical trial is entirely up to you. Even if you sign the informed consent form, you’ll be free to leave the trial at any time.
Qualified participants may receive compensation for time and travel related expenses (including long distance travel). Study-related medications, procedures and examinations are provided at no cost to study participants.
If you agree to participate, you would be expected to follow the rules and instructions to the best of your ability. If you are not able to follow these rules and instructions, you may be asked to withdraw from the study.
In order to provide maximum protection for your health, the study will be under the direct supervision of the study doctor and will be conducted by trained personnel. You will need to provide all information about your current and past health (medical history) at the Screening Visit and at each Follow-Up Visit, including participation in any other research studies. This information is needed to protect your health.
If you have a primary doctor, it is strongly recommended that you inform him/her of your interest to participate in this research study.
Mirum Pharmaceuticals, Inc., based in Foster City, California, is a biopharmaceutical company focused on the development and commercialization of therapies to treat debilitating liver diseases. Mirum currently has two ileal bile acid transporter (IBAT) inhibitors; an investigational medication, volixibat for pediatric and adult liver diseases in development stage and one approved medication.
Mirum works closely with advocates and the patient communities to understand and address the most critical aspects affecting patients and families living with rare liver diseases.
For more information about Mirum, please visit MirumPharma.com.